

Sorbsan Packing is a sterile, non-woven, calcium alginate cavity wound dressing, high in Mannuronic acid and low in Guluronic acid.
As with Sorbsan Flat, he fibres of Sorbsan Packing swell and form Sodium-Calcium alginate gel in contact with wound exudate. The gel:
- Provides a moist wound healing environment1
- Conforms to the contours of the wound2,3
- Atraumatic dressing removal3
- Quick and easy to remove by irrigating with saline (0.9%) solution4
- Promotes haemostasis5,6
- Minimises disruption to newly formed tissue7
Wound types:

INDICATIONS FOR USE
Sorbsan Packing is suitable for the management of larger, wet cavity wounds, such as abscesses and sinuses resulting from:
- Pilonidal sinuses
- Pressure ulcers
- Post-operative wounds
- Fungating lesions
Sorbsan Packing is also suitable for the management of bleeding cavity wounds:
- Following toe-nail avulsions
- Pressure ulcers
- Donor and graft sites
- Traumatic wounds
CONTRA-INDICATIONS
Do not use on patients with a known sensitivity to Sorbsan Packing or any of its components.
Sorbsan Packing is not intended to be used as a surgical sponge.
Sorbsan Packing should not be packed tightly into a wound.
Sorbsan Packing is not intended to control heavily bleeding wounds, alternative measures must be considered in situations incurring excessive loss of blood.
Always refer to the IFU provided with the dressing before use.
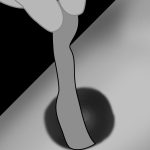
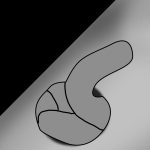
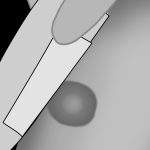
Application


Ordering Information
| Product code | Dressing | Size | Qty | PiP code | NHS code |
|---|---|---|---|---|---|
| 1411 | Sorbsan Packing | 30cm/2g | 5 | 034-8979 | tbc |
We’re changing the way Sorbsan Packing is supplied
Going forward, the Wound Probes provided with Sorbsan Packing and Sorbsan Ribbon will be packaged separately. This will not effect the quality or quantity of the dressings supplied and is solely a change to the packaging. Refer to the Notification of packaging change for more information. Please contact us for all queries and enquiries.
References
1. Clayton, C. An evaluation of Sorbsan Silver for use as an antimicrobial wound dressing. Proceedings of Wounds UK, Harrogate 2009
3. Thomas, S Alginates Update on Alginates. Journal of Woundcare May/June Vol.1. No.1 1992
4. Akrum Allymamod, Use of Calcium Alginate vs Hydrofibre dressing in City and Hackney Community Health Service. Poster presented EWMA 2011
5. Duward,R. Dr. and Clark, M Prof. Novel methods for measuring the ability of wound dressings to contribute to blood coagulation. Poster and presentation EWMA 2012
6. Gove, J. A multicentre study examining the role of calcium alginate dressing in bleeding and chronic wounds Poster presented at EPUAP,EWMA 2012
